Key Takeaways
- Scientists have developed a new material from recycled coffee grounds to clean industrial water.
- This material is highly effective at capturing and removing harmful dyes used in textiles and plastics.
- The substance can be reused multiple times while maintaining its ability to clean pollutants from water.
- Using food waste to create water filters helps the environment by reducing trash and protecting water resources.
- This technology provides a cheaper and more sustainable alternative to current industrial water treatment methods.
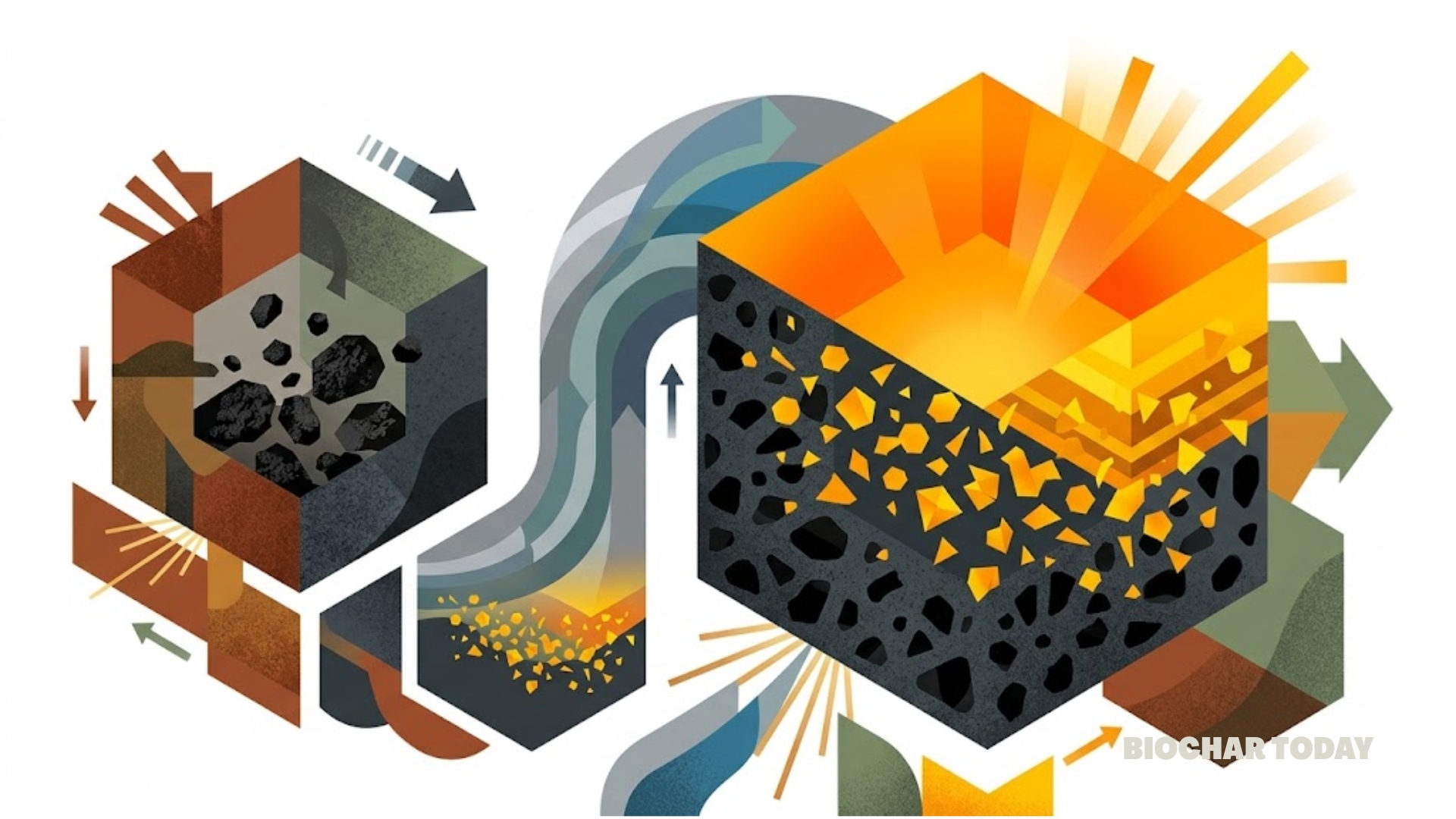
The research published in the journal Nanomaterials Chemistry by authors Tanmay Mandal and Aarav Srinivasan presents a significant advancement in the field of sustainable wastewater treatment. The team successfully engineered a nitrogen-doped biocharBiochar is a carbon-rich material created from biomass decomposition in low-oxygen conditions. It has important applications in environmental remediation, soil improvement, agriculture, carbon sequestration, energy storage, and sustainable materials, promoting efficiency and reducing waste in various contexts while addressing climate change challenges. More and reduced graphene oxide nanocomposite utilizing common coffee waste as a primary precursor. This development addresses the growing environmental concern regarding azo dyes, which are prevalent in the textile, printing, and leather industries. These dyes are notoriously difficult to break down because of their stable chemical bonds and aromatic structures. By repurposing organic waste into a high-performance cleaning agent, the study aligns the principles of the circular economy with advanced materials science to tackle industrial pollution.
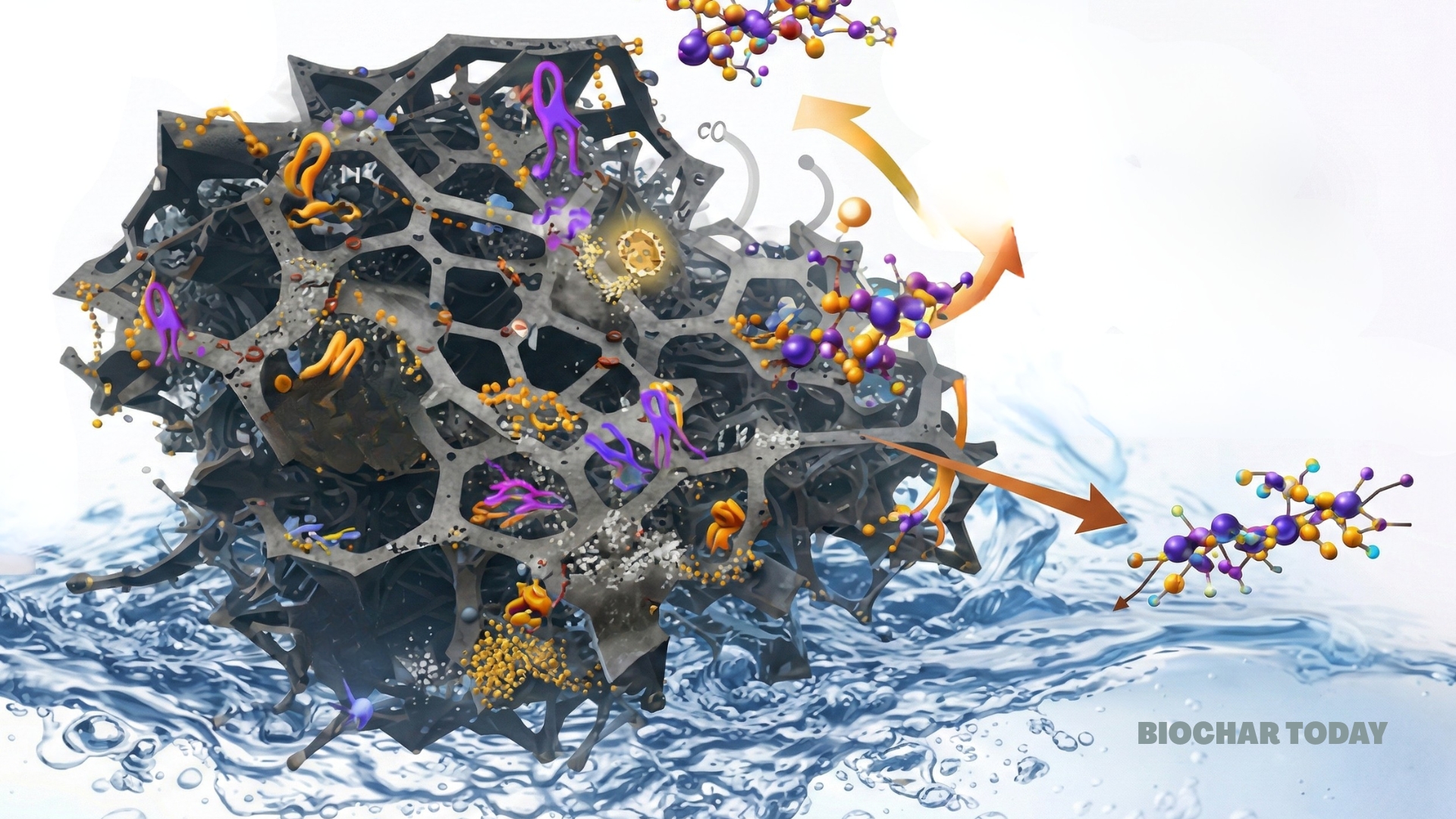
The effectiveness of this new nanocomposite stems from its sophisticated structural design, which the researchers analyzed through various technical lenses. The material exhibits a mesoporous structure, meaning it contains tiny pores that are ideal for trapping dye molecules. Specifically, the analysis showed that the composite has a high specific surface area of 512 square meters per gram. This represents a massive increase in available cleaning space compared to raw biochar produced from coffee waste without these specific enhancements. The average diameter of these pores is approximately 7.4 nanometers, providing ample room for the large molecules found in azo dyes to enter and become stuck to the surface of the adsorbent.
Beyond just physical trapping, the chemical makeup of the material plays a vital role in its performance. Through specialized testing, the authors confirmed that nitrogen was successfully integrated into the carbon network. This nitrogen exists in several different forms, including pyridinic and pyrrolic species, which act as active sites that chemically attract dye molecules. These sites create a strong bond between the adsorbent and the pollutants, especially in acidic conditions. For example, the study found that the material works best at a pHpH is a measure of how acidic or alkaline a substance is. A pH of 7 is neutral, while lower pH values indicate acidity and higher values indicate alkalinity. Biochars are normally alkaline and can influence soil pH, often increasing it, which can be beneficial More of 4, where the surface carries a strong positive charge that naturally pulls in negatively charged dye particles. This combination of physical porosityPorosity of biochar is a key factor in its effectiveness as a soil amendment and its ability to retain water and nutrients. Biochar’s porosity is influenced by feedstock type and pyrolysis temperature, and it plays a crucial role in microbial activity and overall soil health. Biochar More and chemical attraction allows the material to outperform many traditional and biobased alternatives.
One of the most impressive results of the study is the high maximum adsorption capacity recorded during testing with Congo Red, a common industrial dye. The material demonstrated a capacity of 486 milligrams of dye for every single gram of adsorbent used. The data indicates that the dye attaches to the surface in a single, uniform layer, suggesting that the active sites are spread evenly across the material. This high capacity is significant because it means smaller amounts of the coffee-derived composite can clean larger volumes of contaminated water, making the process much more efficient for industrial applications.
Longevity and stability are also crucial factors for any new environmental technology, and the findings here are promising. The researchers tested how well the material holds up after repeated use. Even after five full cycles of cleaning dye from water and then being refreshed, the material retained 88 percent of its original efficiency. This indicates that the structural integrity of the composite is robust and that it does not easily break down during the cleaning process. Such durability is essential for reducing the costs associated with water treatment, as it minimizes the need for frequent replacement of the filtration media.
The study concludes that the synergy between the coffee-waste biochar and the added graphene layers creates a powerful three-dimensional network for water remediation. The incorporation of reduced graphene oxide helps prevent the structure from collapsing and adds further chemical pathways for dyes to be neutralized. By engineering defects into the carbon framework on purpose, the scientists created more “hooks” for pollutants to grab onto. This research proves that intelligent design can transform everyday food waste into an advanced tool for protecting the planet. It offers a clear path forward for industries looking to reduce their environmental footprint while managing waste more effectively.
Source: Mandal, T., & Srinivasan, A. (2025). Sustainable N-doped biochar/rGO nanocomposite from coffee waste for azo dye removal. Nanomaterials Chemistry, 3, 262-273.





Leave a Reply