Article Summary: Mutabazi, et al. (2023) Cr(VI) adsorption on activated carbonActivated carbon is a form of carbon that has been processed to create a vast network of tiny pores, increasing its surface area significantly. This extensive surface area makes activated carbon exceptionally effective at trapping and holding impurities, like a molecular sponge. It is commonly More, sludge derived biocharBiochar is a carbon-rich material created from biomass decomposition in low-oxygen conditions. It has important applications in environmental remediation, soil improvement, agriculture, carbon sequestration, energy storage, and sustainable materials, promoting efficiency and reducing waste in various contexts while addressing climate change challenges. More, and peanut shells derived biochar: Performance, mechanisms during the reuse process and site energy distribution analysis. Journal of Water Process Engineering, In Press. https://doi.org/10.1016/j.jwpe.2023.104679
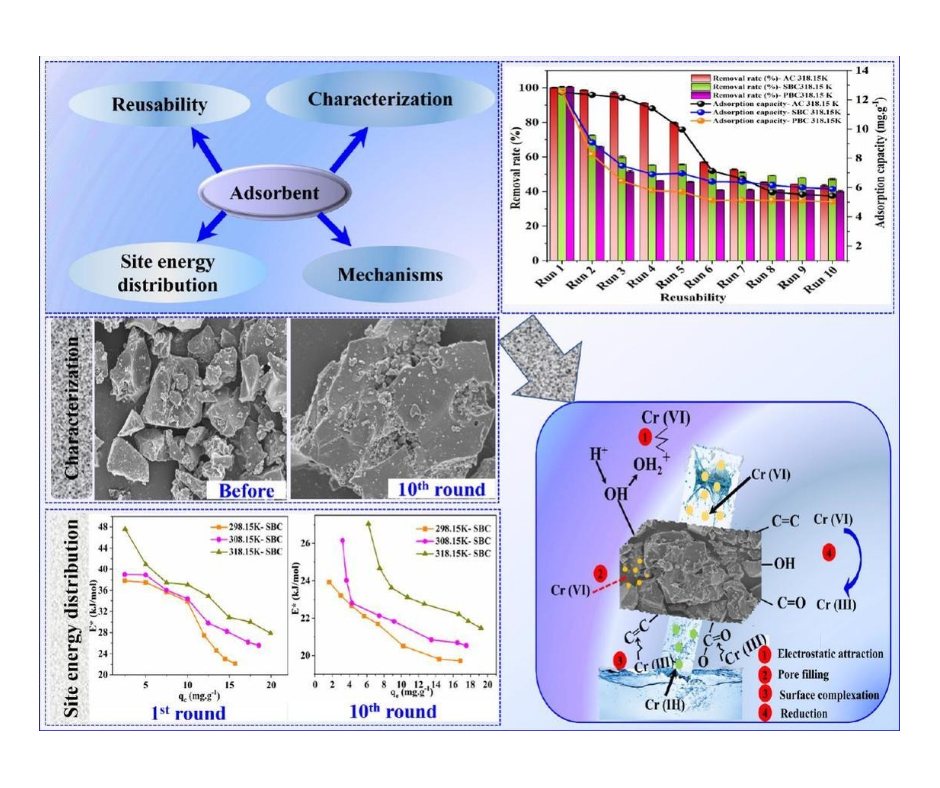
Chromium(VI) (Cr(VI)) contamination in wastewater poses a serious threat to human health and the environment. Finding ways to remove it effectively and sustainably is crucial, and reusable adsorbents offer a promising solution. This study delves into the fascinating world of three such adsorbents: activated carbon, sludge-derived biochar, and peanut shell-derived biochar.
The researchers put these materials through their paces, evaluating their performance in removing Cr(VI) over ten rounds of reuse. While their efficiency declined slightly with each run, the decrease was far from drastic, highlighting their potential for practical application.
But what about the mechanisms behind this performance? The study digs deeper, analyzing the adsorbents’ surface chemistry and energy distribution before and after each reuse cycle. It turns out that the loss of specific functional groups (-OH, C=O, etc.) on the surface plays a role in the decline. However, even with these changes, the adsorbents still managed to hold on to Cr(VI) effectively.
This is because they followed a strategic seating arrangement! The Cr(VI) molecules, like VIP guests, first grabbed the best seats, the high-energy sites on the adsorbent’s surface. As these prime spots filled up, they settled for the less prestigious, but still comfortable, low-energy seats. This unique “site energy distribution” explains why their removal efficiency remained decent even after multiple uses.
Another interesting finding was the role of chromium reduction. Apparently, transforming Cr(VI) to its less harmful form, Cr(III), played a significant role in its removal. This adds another layer to the complex dance of adsorption and detoxification.
This study provides valuable insights into the world of reusable Cr(VI) adsorbents, shedding light on their resilience, surface interactions, and preferred guest seating arrangements. By unlocking these secrets, scientists can design even more effective and sustainable materials for cleaning up contaminated water.
So, next time you hear about wastewater treatment, remember this tale of reusable adsorbents – remarkable materials working tirelessly behind the scenes to protect our environment!




Leave a Reply