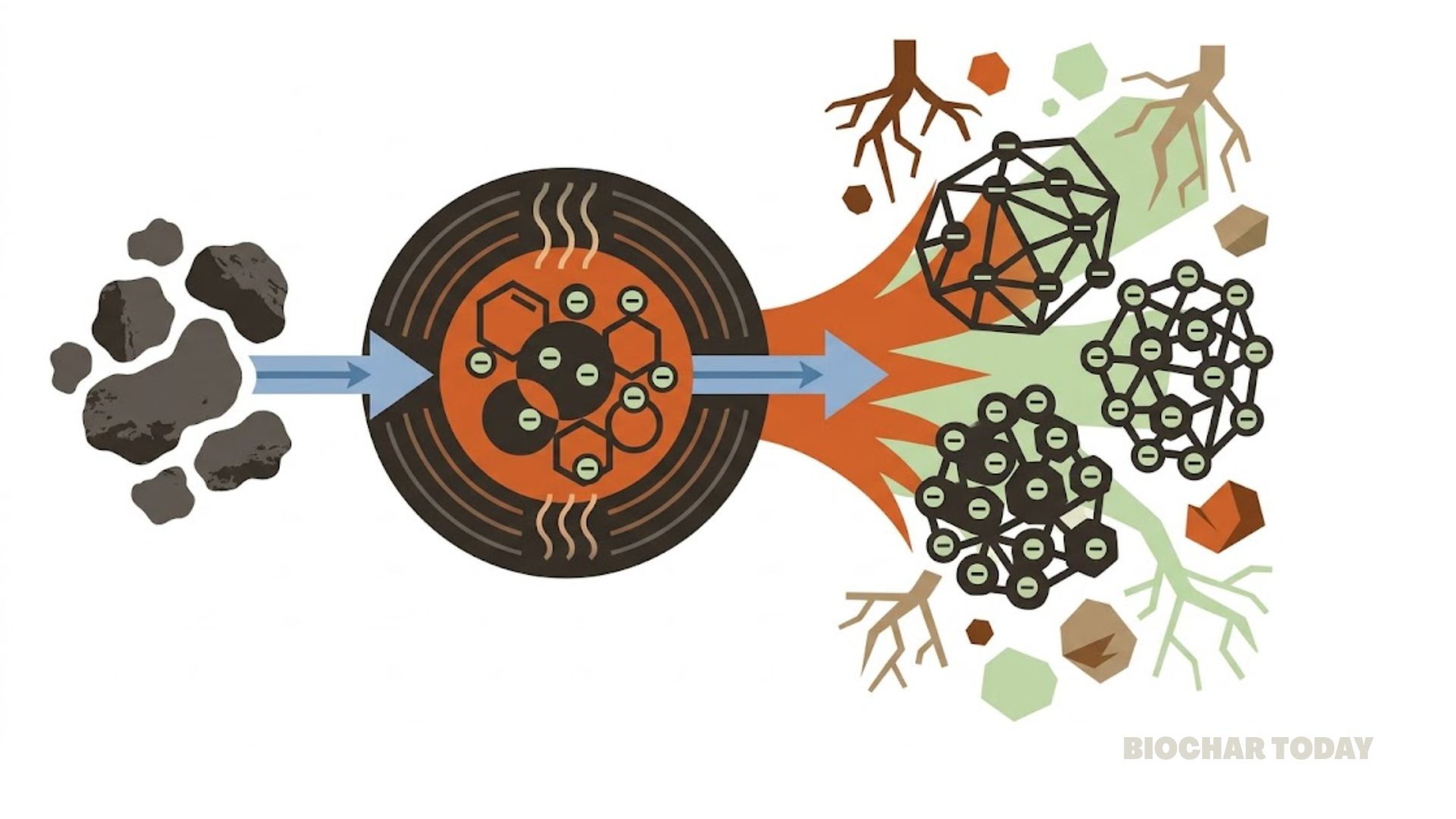
Gasification is a high-temperature, thermochemical process that converts carbon-based materials into a gaseous fuel called syngas and solid by-products. It takes place in an oxygen-deficient environment at temperatures typically above 750°C. Unlike combustion, which fully burns material to produce heat and carbon dioxide (CO2), gasification partially oxidizes the material to maximize the production of syngas.

Why it Matters
- Low Biochar Yield: Gasification is designed to produce syngas, not biochar. The process converts the majority of the carbon in the biomass into gaseous products like carbon monoxide (CO) and carbon dioxide (CO2), resulting in a very low biochar yield—typically around 10%. This makes it an inefficient method for biochar production.
- Product Focus: The primary goal of gasification is to create a gaseous fuel (syngas) which can be used for energy. This is in contrast to other methods like slow pyrolysis, which are specifically designed to maximize the yield of a solid product (biochar).
- Environmental and Economic Trade-offs: While gasification is inexpensive and has a short reaction time, its low biochar yield and emission of greenhouse gases make it less suitable for applications where the main objective is carbon sequestration or producing a solid soil amendment.
Reference
Hansen, V., Müller-Stöver, D., Ahrenfeldt, J., Holm, J. K., Henriksen, U. B., & Hauggaard-Nielsen, H. (2015). Gasification biochar as a valuable by-product for carbon sequestration and soil amendment. Biomass and Bioenergy, 72, 300-308. https://doi.org/10.1016/j.biombioe.2014.10.013